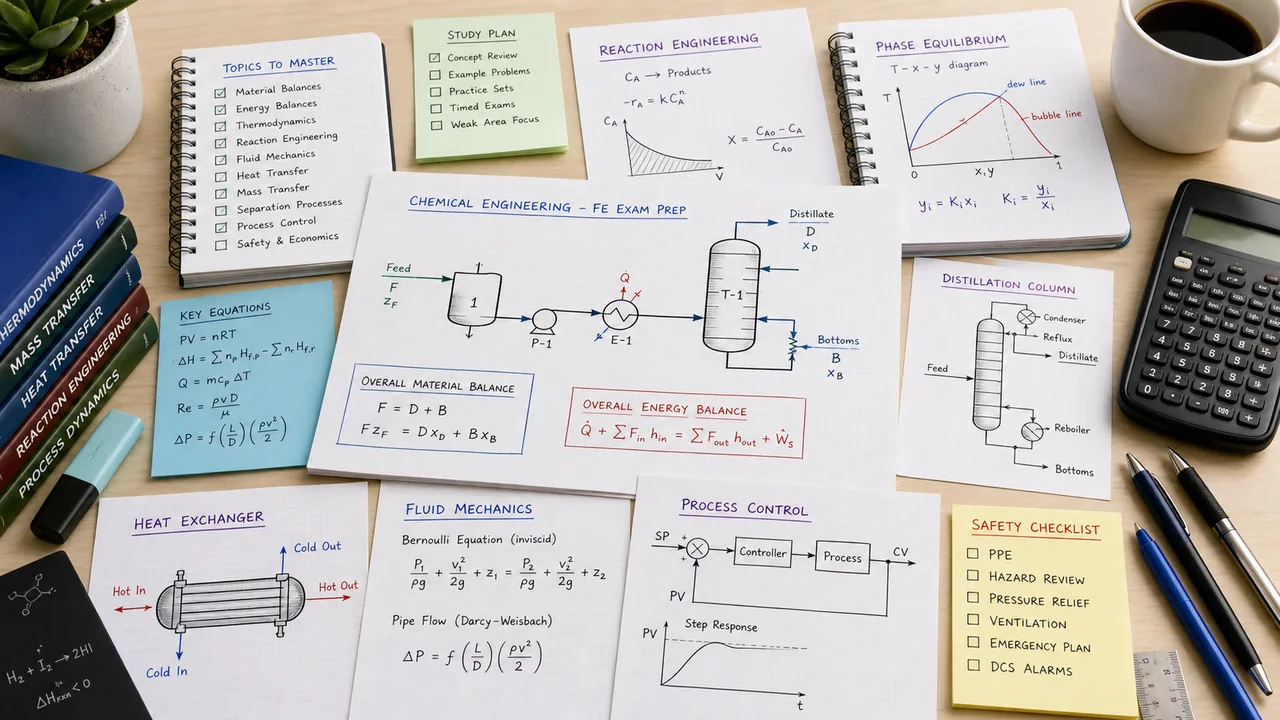
The FE Chemical exam is won in the setup. Material balances, energy balances, thermodynamics, kinetics, and transport problems become much less intimidating once you can identify the system boundary, choose the right relation, and keep units under control. This guide gives you the topic map, the highest-return study order, and a 12-week plan that turns chemical engineering breadth into manageable practice blocks.
FE Chemical Exam at a Glance
- 110 questions in 5 hours 20 minutes (computer-based, year-round)
- 17 topic areas — material/energy balances carry the largest single topic range
- High-return core — balances, thermo, fluids, heat transfer, mass transfer, chemistry, reaction engineering, and process design
- NCEES provides the FE Reference Handbook digitally — no outside notes allowed
- Most successful candidates study 200–300 hours over 8–16 weeks
What Is the FE Chemical Exam and Why Does It Matter?
NCEES administers the computer-based FE exam at Pearson VUE testing centers. You get 110 questions in 5 hours and 20 minutes, plus the digital FE Reference Handbook. Treat the handbook as part of the problem-solving flow: recognize the balance, property, kinetics, or transport pattern, find the right equation or table, and keep the calculator work clean.
Passing the FE exam earns you the designation of Engineer Intern (EI) or Engineer in Training (EIT), depending on your state. From there, after accumulating the required years of professional experience (typically four years under a licensed PE), you become eligible to sit for the PE exam and earn full licensure.
Career Benefits of Passing the FE Exam
- PE licensure pathway: A PE license is required to offer engineering services directly to the public, hold certain senior positions, and sign and seal engineering documents. In chemical engineering, PE licensure is particularly valued in process design, consulting, environmental compliance, and plant operations.
- Competitive advantage: Even before you earn your PE, the EIT credential signals competence and commitment to employers. Many chemical engineering firms in the process industries, oil and gas, pharmaceuticals, and food processing expect new hires to have passed the FE or to pass it within their first year.
- Higher earning potential: Licensed professional engineers consistently earn higher salaries than their unlicensed peers, with many surveys showing a 10–20% premium (Bureau of Labor Statistics).
- Career mobility: Licensure is recognized across all 50 U.S. states and territories through comity agreements, making it easier to work in different jurisdictions.
What Are the 17 Topic Areas on the FE Chemical Exam?
The FE Chemical exam draws from 17 topic areas. NCEES publishes an exam specification that assigns each topic an approximate number of questions out of 110. Below is each topic area with its estimated question count, approximate weight, and what to expect.
| Topic Area | Est. Questions | NCEES Range | Priority |
|---|---|---|---|
| Material & Energy Balances | 10–15 | 10–15 questions | HIGH |
| Chemical Eng. Thermodynamics | 8–12 | 8–12 questions | HIGH |
| Mass Transfer & Separation | 8–12 | 8–12 questions | HIGH |
| Fluid Mechanics/Dynamics | 8–12 | 8–12 questions | HIGH |
| Heat Transfer | 8–12 | 8–12 questions | HIGH |
| Chemistry and Biology | 7–11 | 7–11 questions | MEDIUM |
| Chemical Reaction Engineering | 7–11 | 7–11 questions | MEDIUM |
| Process Design | 7–11 | 7–11 questions | MEDIUM |
| Mathematics | 6–9 | 6–9 questions | MEDIUM |
| Process Control | 4–6 | 4–6 questions | LOW |
| Safety, Health & Environment | 5–8 | 5–8 questions | LOW |
| Probability & Statistics | 4–6 | 4–6 questions | LOW |
| Engineering Sciences | 4–6 | 4–6 questions | LOW |
| Materials Science | 4–6 | 4–6 questions | LOW |
| Economics | 4–6 | 4–6 questions | LOW |
| Solids Handling | 3–5 | 3–5 questions | LOW |
| Ethics & Professional Practice | 3–5 | 3–5 questions | LOW |
Where to Review the Highest-Return FE Chemical Topics
For FE Chemical, the best map is a chain: balances, thermo, transport, reactions, separations, then controls. Use outside resources to repair a weak link, then return to handbook lookup and FE-style practice.
Make every problem obey a balance statement.
Balances are the language of chemical questions. Practice steady-state, transient, recycle, bypass, purge, reactive, and nonreactive setups until the diagram comes before the math.
- Handbook move: know where unit conversions, properties, and balance-related reference data live.
- Practice move: draw system boundaries and label known flow rates before solving.
Track the state, phase, and property model.
Thermo questions often depend on choosing the correct property relationship, phase rule, vapor-liquid equilibrium expression, or energy balance around a device.
- Handbook move: mark property tables, phase behavior, and thermodynamic relationship sections.
- Practice move: write the assumed phase and basis before calculating.
Classify the transport mechanism before grabbing correlations.
Transport problems are lookup-heavy but only after you decide whether the question is pressure drop, heat rate, convection, conduction, diffusion, or mass-transfer driving force.
- Handbook move: group dimensionless numbers, heat-transfer relations, and mass-transfer tools.
- Practice move: check whether the driving force is pressure, temperature, or concentration.
Connect rate law, reactor type, and conversion.
Reaction engineering questions usually become manageable once you identify batch, CSTR, PFR, order, limiting reactant, and whether the problem is asking for rate or size.
- Handbook move: mark kinetics, reactor, and conversion relationships together.
- Practice move: define conversion and limiting reactant before using a design equation.
Use diagrams and cause-effect logic for smaller topics.
Controls, instrumentation, process design, and safety may be lighter than balances or transport, but they are good points when you know symbols, feedback loops, hazards, and equipment choices.
- Handbook move: know the control, instrumentation, and safety reference areas by name.
- Practice move: sketch the loop or process path before answering conceptual items.
Source note: topic names and question ranges should be checked against the current NCEES FE exam page. External resources are optional review aids and may include material outside the FE exam scope.
Below is a detailed look at each topic area and what to expect.
Part 1: Shared Topics (All FE Disciplines)
1. Mathematics (6–9 questions)
Covers calculus (derivatives and integrals), differential equations, linear algebra, vector operations, and numerical methods. You will encounter problems involving partial derivatives, solving first- and second-order ordinary differential equations, matrix operations, Laplace transforms, and vector calculus. The NCEES FE Reference Handbook provides key formulas, but you need to know how to recognize which technique applies and execute it quickly. Mathematics underpins virtually every other topic on the chemical engineering exam, from reaction kinetics (differential equations) to process control (Laplace transforms). Subtopics include:
- Differential and integral calculus
- Ordinary differential equations
- Linear algebra and matrix operations
- Laplace transforms
- Numerical methods (root-finding, numerical integration, Euler’s method)
2. Probability and Statistics (4–6 questions)
Expect questions on probability distributions (normal, binomial, Poisson), measures of central tendency and dispersion, linear regression, hypothesis testing, and confidence intervals. Chemical engineers use statistics in quality control, process optimization, and experimental design. Most problems are straightforward if you understand when to apply each distribution and how to use the standard normal table in the reference handbook. Subtopics include:
- Probability distributions and expected values
- Measures of central tendency and dispersion
- Estimation and hypothesis testing
- Linear regression and correlation
- Confidence intervals and sample size
3. Ethics and Professional Practice (3–5 questions)
Covers the NCEES Model Rules of Professional Conduct, ethical obligations, public safety considerations, and licensure requirements. These are conceptual rather than computational — you will be given scenarios and asked to identify the correct ethical course of action. Read the ethics section in the reference handbook carefully. This is one of the easiest topic areas to score well on with minimal study time, making it essentially free points on exam day. Subtopics include:
- Codes of ethics and professional conduct
- Public health, safety, and welfare obligations
- Licensure laws and regulations
- Professional liability and responsibility
4. Economics (4–6 questions)
Topics include time value of money, present and future worth analysis, annual cost comparisons, benefit-cost analysis, rate of return, breakeven analysis, and depreciation methods. Master the standard factor formulas (P/F, F/P, A/P, A/F, P/A, F/A) and you will handle these efficiently. Chemical engineers routinely use economic analysis to compare process alternatives, evaluate capital projects, and justify equipment purchases. Subtopics include:
- Time value of money and cash flow diagrams
- Present worth, future worth, and annual worth analysis
- Rate of return and benefit-cost analysis
- Depreciation methods (straight-line, MACRS)
- Breakeven analysis
Part 2: Chemical Engineering–Specific Topics
5. Chemistry and Biology (7–11 questions)
A significant topic area that bridges general chemistry with chemical engineering applications. Covers inorganic chemistry (periodic table trends, bonding, molecular structure), organic chemistry (functional groups, nomenclature, basic reaction types), and physical chemistry (electrochemistry, kinetics fundamentals, solutions and colligative properties). Many questions require you to predict reaction products, balance redox equations, or apply concepts like Le Chatelier’s principle. Subtopics include:
- Periodic table trends and chemical bonding
- Stoichiometry: mole ratios, limiting reagents, percent yield
- Chemical equilibrium: Keq, reaction quotient, Le Chatelier’s principle
- Acids and bases: pH, pOH, buffer solutions, Henderson-Hasselbalch equation
- Organic chemistry: IUPAC nomenclature, functional groups, isomerism
- Electrochemistry: standard cell potentials, Nernst equation, Faraday’s laws
- Intermolecular forces: hydrogen bonding, van der Waals, dipole-dipole
6. Material and Energy Balances (10–15 questions)
The bread and butter of chemical engineering. This topic tests your ability to set up and solve material and energy balances on steady-state and transient systems, including systems with recycle, bypass, and purge streams. Expect problems involving multiple units, reactive systems with extent of reaction, combustion processes, and combined material and energy balances. This is the foundation on which chemical engineering thermodynamics, reaction engineering, and separation processes are built. Subtopics include:
- Steady-state material balances on single and multi-unit systems
- Degree-of-freedom analysis
- Reactive systems: conversion, yield, selectivity, extent of reaction
- Recycle, bypass, and purge stream calculations
- Combustion: excess air, theoretical air, flue gas composition
- Energy balances: sensible heat, latent heat, heat of reaction
- Combined material and energy balances
- Transient (unsteady-state) material balances
7. Chemical Engineering Thermodynamics (8–12 questions)
The most conceptually demanding topic on the exam. Covers the first and second laws of thermodynamics, thermodynamic properties of pure substances and mixtures, equations of state (ideal gas, van der Waals, Peng-Robinson), phase equilibria (VLE, LLE), fugacity and activity coefficients, Raoult’s law and modified Raoult’s law, power cycles, refrigeration cycles, and chemical equilibrium. Subtopics include:
- First law: closed systems (Q − W = ΔU) and open systems (SFEE)
- Second law: Carnot efficiency, entropy generation, irreversibility
- Thermodynamic properties: steam tables, property diagrams (T-s, P-h)
- Equations of state: ideal gas, cubic equations (van der Waals, SRK, Peng-Robinson)
- Phase equilibria: Raoult’s law, modified Raoult’s law, Henry’s law
- Fugacity, fugacity coefficients, and activity coefficients
- VLE calculations: bubble point, dew point, flash calculations
- Power and refrigeration cycles: Rankine, Brayton, vapor-compression
- Chemical equilibrium: equilibrium constant, Gibbs free energy
8. Fluid Mechanics (5–8 questions, 5–8%)
Covers fluid properties (density, viscosity, surface tension), fluid statics and manometry, Bernoulli’s equation, the continuity equation, pipe flow with friction losses (Darcy-Weisbach and Moody diagram), Reynolds number, flow through packed beds (Ergun equation), fluidization, and pump sizing. Subtopics include:
- Fluid statics: pressure variation, manometers, forces on submerged surfaces
- Bernoulli’s equation and the mechanical energy balance
- Internal flow: laminar vs. turbulent, Darcy-Weisbach, Moody diagram
- Minor losses: valves, fittings, expansions, contractions
- Flow through packed beds: Ergun equation, pressure drop
- Fluidization: minimum fluidization velocity
- Pump sizing: head, power, efficiency, NPSH, system curves
- Non-Newtonian fluid behavior and rheology
9. Heat Transfer (5–8 questions, 5–8%)
Covers conduction (Fourier’s law, thermal resistance, composite walls, cylinders), convection (Newton’s law of cooling, forced and natural convection correlations), radiation (Stefan-Boltzmann law, emissivity, view factors), and heat exchanger design and analysis (LMTD and effectiveness-NTU methods). Heat exchangers are central to chemical process engineering — from reboilers and condensers to reactor cooling systems. Subtopics include:
- Steady-state conduction: plane wall, cylinder, sphere, thermal circuits
- Fins: effectiveness, efficiency, and extended surfaces
- Forced and natural convection correlations (Re, Nu, Pr)
- Radiation: blackbody, graybody, view factors
- Heat exchangers: LMTD method, effectiveness-NTU method, fouling factors
- Boiling and condensation fundamentals
10. Mass Transfer and Separation (8–12 questions)
One of the defining topics of chemical engineering. Covers molecular diffusion (Fick’s law), convective mass transfer, interphase mass transfer, and the major separation operations: distillation, absorption, stripping, extraction, membrane separation, and drying. Expect problems on McCabe-Thiele diagrams, relative volatility, Fenske equation, Underwood equation, and equilibrium stage calculations. Subtopics include:
- Molecular diffusion: Fick’s first and second laws, diffusion coefficients
- Convective mass transfer: mass transfer coefficients, Sherwood number
- Interphase mass transfer: two-film theory, overall coefficients
- Distillation: VLE, relative volatility, McCabe-Thiele, minimum reflux
- Fenske equation for minimum stages at total reflux
- Absorption and stripping: operating lines, minimum solvent ratios
- Liquid-liquid extraction fundamentals
- Membrane separation: permeability, selectivity
11. Chemical Reaction Engineering (7–11 questions)
Covers reactor design and analysis for batch reactors, continuous stirred-tank reactors (CSTRs), plug flow reactors (PFRs), and packed bed reactors (PBRs). Topics include rate laws, rate constants, reaction order, Arrhenius equation, conversion, selectivity, yield, and reactor sizing. You must be comfortable deriving design equations for ideal reactors, handling multiple reactions, and comparing CSTR and PFR performance. This topic is unique to chemical engineering and is a defining feature of the FE Chemical exam. Subtopics include:
- Rate laws: power law, elementary reactions, rate constants
- Reaction order determination from experimental data
- Arrhenius equation: activation energy and pre-exponential factor
- Batch reactor design: integrated rate equations
- CSTR design equation and sizing
- PFR design equation and sizing
- Levenspiel plots: graphical comparison of CSTR and PFR volumes
- Multiple reactions: selectivity, yield, series and parallel reactions
- Catalysis fundamentals and packed bed reactors
- CSTRs in series, PFR-CSTR combinations
12. Process Design (7–11 questions)
Covers process flow diagrams (PFDs), piping and instrumentation diagrams (P&IDs), equipment selection and sizing, cost estimation, profitability analysis, and process optimization. Chemical engineers are expected to read and interpret PFDs and P&IDs, apply scaling rules for equipment costs, and evaluate project profitability. Subtopics include:
- PFD and P&ID interpretation and symbols
- Equipment sizing: distillation columns, heat exchangers, reactors, pumps
- Capital cost estimation: purchased equipment cost, Lang factors, six-tenths rule
- Operating cost estimation: utilities, raw materials, labor
- Profitability measures: NPV, ROI, payback period
- Process optimization and debottlenecking
13. Process Control (4–6 questions)
Covers feedback and feedforward control, transfer functions, block diagrams, stability analysis, PID controllers, and controller tuning. Process control is essential in chemical plants for maintaining product quality, safety, and efficiency. Expect questions on first-order and second-order system response, gain and time constants, and closed-loop stability. Subtopics include:
- Laplace transforms and transfer functions
- Block diagram algebra: series, parallel, feedback loops
- First-order and second-order system response
- PID control: proportional, integral, derivative actions
- Stability analysis: Routh criterion, Bode plots
- Controller tuning methods
- Feedforward and cascade control
14. Safety, Health, and Environment (5–8 questions)
Covers hazard identification and analysis, process safety management, environmental regulations, and industrial hygiene. This topic is particularly relevant to chemical engineering because of the hazardous materials and high-energy processes involved. These questions tend to be conceptual and scenario-based. Subtopics include:
- Flammability limits: LEL, UEL, flash point, autoignition temperature
- Toxicology: TLV, PEL, LC50, LD50, exposure limits
- Hazard analysis methods: HAZOP, FMEA, fault tree, event tree
- Pressure relief systems: relief valve sizing, rupture disks
- Environmental regulations: Clean Air Act, Clean Water Act, RCRA
- Material Safety Data Sheets (SDS) interpretation
- Inherently safer design principles
Which Topics Should You Prioritize?
Not all 17 topics carry equal weight. A strategic study plan focuses your limited time where it will earn the most points. Here is a three-tier priority system based on the NCEES question weights:
The Big Four (28–44 questions, ~25–40% of the exam)
- Material and Energy Balances — the foundation of chemical engineering; weaknesses here cascade into every other ChemE topic
- Chemical Engineering Thermodynamics — the most conceptually demanding topic; rewards thorough preparation
- Mass Transfer and Separation — distillation, absorption, and extraction are uniquely ChemE and heavily tested
- Fluid Mechanics/Dynamics and Heat Transfer — transport questions reward fast formula lookup and clean units
Material/Energy Balances is the largest single topic range, and the transport/thermo/separations cluster is close behind. If you can consistently answer these correctly, you have built a commanding foundation for passing.
Tier 2: Medium-Weight Topics (24–38 questions, ~22–35% of exam)
After building that core, focus on these topics which round out the technical exam:
- Chemistry and Biology (7–11 questions) — draws on your general and organic chemistry fundamentals; many questions are straightforward for those with a strong chemistry background
- Mathematics (6–9 questions) — underpins thermodynamics, reaction engineering, and process control; differential equations and linear algebra are especially important
- Fluid Mechanics (5–8 questions) — pipe flow, pump sizing, packed beds, and the mechanical energy balance
- Heat Transfer (5–8 questions) — conduction, convection, radiation, and heat exchanger design with LMTD and effectiveness-NTU methods
Together, these high-return technical topics cover most of the places where Chemical candidates can gain or lose time.
Tier 3: Lower-Weight Topics (20–36 questions, ~18–33% of exam)
Finally, review these topics which each carry 4–6 questions:
- Probability and Statistics (4–6 questions) — straightforward with formula lookup from the reference handbook
- Ethics and Professional Practice (4–6 questions) — conceptual, minimal study required; essentially free points
- Engineering Economics (4–6 questions) — formulaic, high score potential with brief review of factor formulas
- Process Design and Economics (4–6 questions) — PFDs, P&IDs, cost estimation, and profitability analysis
- Process Control (4–6 questions) — transfer functions, block diagrams, and PID controllers are learnable in focused study sessions
- Safety, Health, and Environment (4–6 questions) — mostly conceptual; many questions can be answered with sound engineering judgment and basic safety knowledge
While individually these topics carry fewer questions, together they still account for roughly 20–36 questions. Ethics, Engineering Economics, and Safety in particular offer a strong return on a small time investment because the questions tend to be conceptual or formula-driven with short solution paths.
How Should You Structure Your 12-Week Study Plan?
Most successful candidates spend 200–350 hours preparing over 8 to 16 weeks. Here is a suggested 12-week plan tailored to the FE Chemical exam:
- Weeks 1–2: Take a diagnostic practice exam to identify your strengths and weaknesses. Review Mathematics, Probability and Statistics, Engineering Economics, and Ethics. These foundational and lower-weight topics warm up your problem-solving skills and build confidence early. Download the FE Reference Handbook and begin studying with it open so that navigating it becomes second nature.
- Weeks 3–4: Deep dive into Material and Energy Balances. Practice single-unit and multi-unit steady-state balances, recycle and bypass streams, combustion stoichiometry, and combined material and energy balances. Master the degree-of-freedom analysis. This is the cornerstone of chemical engineering and the foundation for the rest of the exam.
- Weeks 5–6: Study Chemistry and Chemical Engineering Thermodynamics intensively. Work through stoichiometry, chemical equilibrium, acid-base chemistry, electrochemistry, and organic chemistry nomenclature. Then tackle thermodynamics: first and second law applications, equations of state, property diagrams, and steam table lookups.
- Weeks 7–8: Continue Chemical Engineering Thermodynamics with phase equilibria (VLE, Raoult’s law, modified Raoult’s law, Henry’s law), fugacity and activity coefficients, and power and refrigeration cycles. Then move to Chemical Reaction Engineering: rate laws, reactor design equations (batch, CSTR, PFR), conversion calculations, and Arrhenius kinetics.
- Weeks 9–10: Cover the three transport topics together: Fluid Mechanics (pipe flow, Bernoulli, pumps, packed beds), Heat Transfer (conduction, convection, radiation, heat exchangers), and Mass Transfer and Separation (diffusion, distillation, absorption, extraction). Focus on the most commonly tested problem types: Darcy-Weisbach for pipe flow, LMTD for heat exchangers, and McCabe-Thiele for distillation.
- Weeks 11–12: Review Process Design and Economics, Process Control, and Safety, Health, and Environment. Practice PFD and P&ID interpretation, cost estimation, transfer functions, block diagram algebra, PID control concepts, flammability limits, HAZOP methodology, and environmental regulations. Take two or more full-length timed practice exams. Review every question you get wrong and revisit weak areas.
What Study Tips Will Help You Pass?
- Learn the reference handbook: The NCEES FE Reference Handbook is provided digitally during the exam. You cannot bring your own notes, so become familiar with where formulas are located and how they are presented. For the chemical engineering exam, pay particular attention to the steam tables, VLE equations, reactor design formulas, and mass transfer correlations.
- Use an approved calculator: Only NCEES-approved calculators are permitted. The TI-36X Pro, Casio FX-115 series, and TI-30X series are popular and reliable choices. Practice with your chosen calculator extensively — the matrix solver is particularly valuable for material balance problems with multiple unknowns.
- Practice under timed conditions: You have roughly 2.9 minutes per question. Build your pacing instincts by taking full practice exams with a timer. If a problem is taking too long, flag it and move on — you can return to flagged questions later.
- Do not leave questions blank: There is no penalty for guessing. If you are stuck, eliminate what you can, choose an answer, flag it, and move on. An educated guess is always better than a blank response.
- Focus on units and basis selection: Chemical engineering problems often require choosing a basis (e.g., 100 mol of feed, 1 hour of operation) and tracking units carefully through multi-step calculations. A wrong basis or a unit conversion error can lead you to a distractor answer. Carry your units through every calculation.
- Leverage your chemistry background: The FE Chemical exam includes a dedicated Chemistry section that other FE disciplines do not have. If you have a strong chemistry foundation, this is an area where you can pick up easy points. Review stoichiometry, organic chemistry nomenclature, and electrochemistry early in your preparation.
- TI-36X Pro Scientific Calculator — the most popular NCEES-approved calculator for the FE exam. Its matrix solver is invaluable for material balance problems.
- NCEES FE Reference Handbook (print copy) — study with a physical copy daily so you can navigate the digital version quickly on exam day.
Final Thoughts
The FE Chemical Engineering exam is challenging, but it is absolutely passable with a structured study plan and consistent effort. Focus on Material/Energy Balances first, then build outward to thermodynamics, fluids, heat transfer, mass transfer, chemistry, reaction engineering, and process design. Finish with the compact topics that offer quick points with focused practice. Become fluent with the reference handbook, take plenty of practice exams under realistic conditions, and manage your time carefully on exam day. Every hour you invest in preparation brings you one step closer to your PE license and the career opportunities that come with it.
How to Pass the FE Chemical Exam • FE Chemical Practice Problems • Best FE Exam Prep Books • 🔢 Calculator Guide • ✅ Exam Day Checklist • Guide for Returning Engineers
Frequently Asked Questions
How many questions are on the FE Chemical exam?
The FE Chemical exam has 110 multiple-choice questions answered over 5 hours and 20 minutes. The exam is computer-based and offered year-round at Pearson VUE testing centers across the United States and internationally.
What are the highest-weight topics on the FE Chemical exam?
Material/Energy Balances is the largest FE Chemical topic range at 10–15 questions. The next highest-return group includes Thermodynamics, Fluid Mechanics/Dynamics, Heat Transfer, and Mass Transfer and Separation at 8–12 questions each, followed by Chemistry/Biology, Reaction Engineering, and Process Design at 7–11 questions each.
How long should I study for the FE Chemical exam?
Plan for 200–300 hours of study over 8 to 16 weeks. A 12-week study timeline works well for most candidates. Prioritize balances, thermodynamics, transport, separations, chemistry, reaction engineering, and process design first, then cover the compact supporting topics.
What reference materials are available during the FE Chemical exam?
NCEES provides a searchable digital copy of the FE Reference Handbook on your exam computer. No outside notes or materials are allowed. The handbook contains key formulas, steam tables, and property data. Study with it daily so you can navigate it quickly on exam day.
Disclaimer: This guide is an independent educational resource and is not affiliated with, endorsed by, or sponsored by NCEES. The “Fundamentals of Engineering” exam, “FE” exam, and “NCEES” are trademarks of the National Council of Examiners for Engineering and Surveying. Exam specifications and content are subject to change; always refer to the official NCEES website for the most current information.
Ready to Start Practicing?
Practice the Chemical workflow: define the system, choose the relation, track units, and review the distractor logic when a setup goes sideways. FE Test Prep pairs Chemical questions with step-by-step solutions, key signals, Quick Formula Reference support, and calculator help so each miss becomes a cleaner next attempt.
Start the free FE Chemical diagnostic
Enter your email for FE Chemical study updates, then jump into the free 20-question diagnostic. No account needed.